What Math Do You Need to Know for Astrophysics
Learn cardinal MCAT topics almost MCAT math, plus practice questions and answers

(Note: This guide is part of our MCAT Physics series.)
Table of Contents
Office 1: Introduction
Function 2: Estimating and rounding numbers
a) Multiplication
b) Division
c) Example problem
Function 3: Logarithms
a) Logarithm rules
b) Common applications of logarithms
c) Example trouble
Part 4: Strategies for eliminating reply choices
a) Significant figures
b) Exponents
c) Example problem
Function 5: Fractions
a) Addition and subtraction
b) Multiplication and sectionalisation
c) Application: sparse lens equation
d) Example problem
Role six: Passage-based questions and answers
Part seven: Standalone questions and answers
Part i: Introduction
While the MCAT does non permit the apply of a calculator, the test still contains questions that will require mathematical calculations—particularly in the context of physics and chemistry. Fine-tuning your math skills tin can thus significantly improve your power to piece of work through these test questions.
While many of these math skills include basic addition, subtraction, multiplication, and addition, this section contains a few additional concepts that are of import to castor up on for the MCAT, including logarithm rules and manipulating fractions. At the end of this guide, we take included several do problems for you to practice with.
Let'south dive in!
Function two: Estimating and rounding numbers
Estimating and rounding numbers are useful skills on the MCAT, as they can assist you calculate problems more than effectively. This section contains important reminders to guide you through estimating and rounding.
a) Multiplication
Suppose we're trying to multiply 6.84 x 2.25. With no reckoner available, how can we all-time guess this answer in a brusque corporeality of fourth dimension?
When multiplying numbers with many nonzero digits, rounding can requite u.s. more digestible numbers to work with. Rounding these numbers to one decimal identify or a whole number can greatly simplify our mathematical calculations.
To determine how much to circular numbers, it may be helpful to kickoff look at the answer choices that are presented. If the answer choices are closer together, so it would be better for us to round to one fewer digit to go along our calculations more accurate. If our reply choices are further autonomously, and so we tin can get away with rounding more.
To keep our calculations accurate, we can likewise recoup for our rounding for each digit. If we decide to round one number up, nosotros should circular the other number downwardly, and so that nosotros stay closer to the true value. For instance, if we round 6.84 to half dozen.8, then we should round 2.25 to 2.3.
Suppose we chose to round 2.25 down to ii.ii instead. How would this compare to the correct reply and the answer provided past using two.3?
$$half dozen.84 \times 2.25 = xv.39 $$ $$half-dozen.8 \times 2.iii = 15.64 \rightarrow 15.6 $$ $$ 6.8 \times 2.2 = fourteen.96 \rightarrow 15.0 $$
Our calculation is further from the right answer when we use 2.two. By compensating for rounding and using the digit two.3, we become a result closer to the actual answer of 15.39.
b) Segmentation
Estimating and rounding in division problems is like to what we do for multiplication problems. As with multiplication, we start check the answer choices to determine how much we should round.
In division, however, we make dissimilar adjustments when nosotros compensate for our rounding. To go on our numbers proportional to each other, both the dividend and divisor should exist shifted in similar directions.
Say nosotros desire to separate 19.58 by iv.67. If nosotros make up one's mind to round 19.58 upward to 20, so we should likewise round 4.67 up to 5. Doing so will give united states a result of 4, which is close to the actual 4.19.
c) Example problem
Let's work through an example of a mathematical trouble that is likely to be found on the MCAT.
A student adds 6.19 grams of solid hydrochloric acid to 0.0500 50 of water. What is the molarity of the resulting solution?
Molarity is given in moles per liter. To calculate the molarity of this solution, we will showtime by determining the number of moles of muriatic acid (HCl). Outset, we calculate the molar mass of HCl.
$$ one.008 \space ^g/_{mol} \space + 35.45 \space ^g/_{mol} = 36.458 \space ^g/_{mol} $$
Then, we employ this tooth mass to convert the mass of HCl to moles. We can round both numbers down to simplify our calculation.
$$ 6.19 \space g \infinite HCl \div (36.458 \infinite ^g/_{mol} \infinite HCl)$$ $$ 6 \space one thousand \space HCl \div (36 \infinite ^k/_{mol} \space HCl) = 0.1667 \infinite mol \space HCl $$
We then divide this value by the book of the solution to decide molarity.
$$ 0.1667 \space mol \space HCl \space \div 0.0500 50 = 3.33 M \infinite HCl $$
The approximate molarity of the solution is thus calculated to be iii.33M HCl.
Part 3: Logarithms
a) Logarithm rules
Logarithms are another mathematical concept likely to appear on the MCAT. These functions are the inverse of an exponential part, so they piece of work by like rules. Hither are some important rules and operations to remember when manipulating logarithms:
-
The log of 1 is ever 0.
-
The log of the base of operations number is always 1.
-
So long every bit the base numbers are the same, the log of two factors equals the sum of the log of the individual factors.
$$ log_n(x\times y) = log_n(x) + log_n(y) $$
-
Likewise, the log of a fraction equals the difference between the log of the dividend and the log of the divisor.
$$ log_n(x \div y) = log_n(x) - log_n(y) $$
b) Common applications of logarithms
One mutual logarithm yous might see is with base of operations "e." These logarithms are known as natural logarithms and are denoted as ln().
Logarithms with base 10 are as well very common. (You should be very familiar with how to manipulate and approximate logarithms with base 10.) For the MCAT, it is of import to note that the prefix p signifies –log. For instance, pH = -log[H+], and pOH = -log[OH-].
Decibels are another common application of logarithms in the MCAT. Decibels (dB) are a measure out of audio level and can be calculated through the post-obit equation:
$$ dB = 10log(\frac{I}{I_0}) $$ $$ where \space I = \mbox{intensity of the sound, in } \frac{W}{grand^2}$$ $$ I_0 = \mbox{threshold of the lowest audible audio to a healthy homo, equal to } 10^{-12} \frac{Due west}{m^2}$$
The Henderson-Hasselbalch equation is a common application of logarithms on the MCAT. This equation allows united states of america to conduct buffer calculations and tin can be derived from the equilibrium abiding expression for the dissociation of a weak acid. The Henderson-Hasselbalch equation is as follows:
$$ pH = pK_a + log(\frac{[A^-]}{HA})$$
c) Example problem
Let'south piece of work through an application of using logarithms.
The pH of a solution of HF (pKa = 3.2) is 4.8. What is the ratio of acrid to conjugate base in the HF solution?
The Henderson-Hasselbalch equation relates all of these values. We can use it to solve for the ratio.
$$ pH = pK_a + log(\frac{[A^-]}{HA})$$
Nosotros showtime by isolating our ratio on one side of the equation.
$$ pH - pK_a = log(\frac{[A^-]}{[HA]})$$ $$ -one \times (pH - pK_a) = -1 \times log(\frac{[A^-]}{[HA]}) $$ $$ pK_a - pH = log(\frac{[HA]}{[A^-]})$$
From here, nosotros can plug in our pKa and pH values.
$$ iii.2 - 4.viii = log(\frac{[HA]}{[A^-]})$$ $$ -1.6 = log(\frac{[HA]}{[A^-]})$$
To obtain the ratio, nosotros must apply the inverse of the logarithm function – the exponential office. Since log() is the notation for base 10, we can heighten the number 10 to the values present on both sides of the equation. Raising the number 10 to a logarithm with its ain base volition effectively "cancel" the logarithm function on that side of the equation.
$$ 10^{-1.6} = \frac{[HA]}{[A^-]}$$
The ratio of HF to its conjugate base of operations F - in this solution is thus calculated to be ten -1.half dozen . We tin further judge this value by determining a range that this value should be on. While we do not know what the exact value of 10-1.6 is, we exercise know the exact values of 10-1 and ten-2:
$$ 10^ii \lt 10^{-ane.half-dozen} \lt 10^{-ane} $$ $$ \frac{1}{100} \lt x^{-i.vi} \lt \frac{1}{10}$$ $$ 0.01 \lt 10^{-1.6} \lt 0.one $$
Thus, nosotros would expect for an answer option that lies somewhere between 0.01 and 0.1.
Part 4: Strategies for eliminating answer choices
When working through calculation problems, it's probably all-time to endeavour calculating an exact answer, then checking to meet if your answer matches an respond option. However, when in a time crunch, call up that you simply demand to pick ane out of four answer choices! Below we will outline some tips to help ameliorate your speed and accuracy in eliminating respond choices.
Scientific annotation is a method of writing numbers with a significand and exponent.
-
Significands are any real number whose accented value is between 1 and x, only not including 10.
-
The exponent is base of operations 10 and tin can be whatever whole number (i.east., negative, zero, or positive).
a) Significant figures
Often, nosotros can eliminate answer choices using pregnant figure calculations. When reply choices are written in scientific notation, focusing on the number of significant figures in the significand is useful.
Significant figures convey the certainty of a measurement based on its measuring musical instrument. They are necessary to consider in calculations. The post-obit rules can be used make up one's mind significant figures:
-
All numbers betwixt the showtime nonzero digit on the left and the last nonzero digit on the right are significant.
-
When there is a decimal in the number, zeroes to the right of the last nonzero digit are meaning. If at that place is no decimal, then those zeroes are not significant. (For example, the number 4300.00 has six pregnant figures, simply the number 4300 has two significant figures.)
-
Leading zeroes, or zeros preceding the outset nonzero digit, are not significant. (For example, the number 0.x has 2 significant figures.)
Conducting calculations with significant figures also requires specific rules.
-
For calculations with addition and subtraction, brainstorm by noting the position of the leftmost decimal point. From in that location, nosotros proceed with computing the problem and continue equally many digits as we tin can until the very end of our adding. Afterwards we have obtained this result, we circular the calculated answer to the decimal place determined in the first stride.
-
For calculations with multiplication and sectionalisation, first identify which cistron, divisor, or dividend has the least number of pregnant digits. We then calculate the problem—once more, keep as many digits until the very end. Finally, nosotros circular the calculated answer to the number of significant digits of the factor, divisor, or dividend determined in the commencement step.
b) Exponents
Exponents are some other method to eliminate answer choices. Because they are the inverse of the logarithm part, exponential functions follow like rules:
$$ A^0 = 1 $$ $$ A^ten \times A^y = A^{x+y} $$ $$ \frac{A^x}{A^y} = A^{10-y}$$ $$ (A^x)^y = A^{x\times y}$$ $$ (\frac{A}{B})^x = \frac{A^x}{B^10}$$ $$ A^{-x} = \frac{one}{A^ten}$$
When respond choices are written in scientific notation, the base number of the exponent is e'er 10. For questions that are addressing very modest or very large quantities, we can check the exponent of this base of operations number 10 to eliminate answer choices. For instance, if a question asks for the number of molecules in a few moles of substance, we should expect a very large number. In this instance, the answer will more than likely contain an exponent raised to the 23rd power rather than the 5th ability.
c) Example problem
Let's utilise this knowledge with a practice problem.
Summate the molarity of 0.424 mol HI in 2.1 L of aqueous solution.
Let's get-go identify which number has the fewest number of significant digits. The solution volume has fewer significant figures than the number of moles of HI. Because the volume has 2 meaning figures, our final answer will as well accept 2 significant figures.
We can now summate the problem, without rounding until the very cease.
$$ 0.424 \mbox{ mol HI} \div 2.i \space L = 0.201 \infinite M$$
We then rewrite this respond in scientific annotation. First, we adjust the significand and multiply by the proper base ten exponent. Since we movement the decimal over one identify to the correct, we multiply by 10-1.
$$ 0.2019 M \rightarrow 2.019 \times 10^{-1} \space Thou$$
Nosotros finally round this calculated answer to 2 meaning figures to obtain our last answer.
$$ 0.2019 One thousand \rightarrow two.0 \times ten^{-one} \space Chiliad$$
When eliminating respond options, we can be certain to eliminate whatever reply options with large positive numbers in the exponent.
Office 5: Fractions
The MCAT may crave y'all to solve equations that deal with fractions. Y'all may often see fractions applied in the context of the sparse lens equation and solving optical systems.
a) Add-on and subtraction
When adding and subtracting fractions, we apply our operations to the numerators and continue the denominator. We must thus remember to detect a mutual denominator before proceeding with our calculations. This is of import in keeping a fraction balanced.
To pick a common denominator, we expect at the least common multiple of the denominators we are working with. A least common multiple refers to the smallest number that both denominators are a factor of.
For example, if we want to add the post-obit fractions:
$$ \frac{2}{3} + \frac{i}{ix}$$
Nosotros must find the smallest number that both 3 and 9 tin be a factor of. In this case, the least common multiple is 9, since both 3 and 9 are factors of 9.
From here, we rewrite 2/3 equally a fraction with a denominator of 9. To maintain the fraction'south proportionality, we must apply the same operation to the pinnacle and lesser. The denominator must be multiplied by 3 to produce the desired result of 9, so we multiply the acme by iii to get 6. Afterwards converting 2/three to half dozen/9, we can go on with adding.
$$ \frac{6}{9} + \frac{1}{9} = \frac{6+ane}{nine} = \frac{7}{ix}$$
b) Multiplication and partitioning
With multiplication, nosotros multiply the numerators and denominators to obtain our result. In this case, we don't need to find a common denominator before multiplying. Sometimes, nonetheless, we can simplify our calculations before multiplying. First, we check to see if either factor can exist simplified in itself. Then, we check to run across if the numerators of any fraction can be simplified with denominators of other fractions, or vice versa. Afterwards, we proceed with calculating.
For instance, if we're multiplying the following fractions:
$$ \frac{ii}{7} \times \frac{fourteen}{16}$$
We tin can first by simplifying 14/16 to 7/8. Our multiplication problem can then be rewritten every bit:
$$ \frac{two}{7} \times \frac{7}{viii}$$
From here, we can simplify with the numerators of one fraction that divides the denominator of another. Notation that the number 7 divides both the denominator of the showtime fraction and the numerator of the second fraction.
Additionally, the number 2 divides both the numerator of the first fraction and the denominator of the 2d fraction.
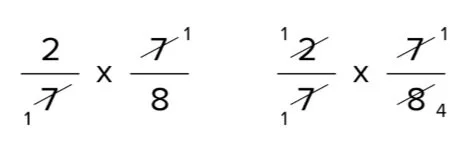
After simplifying these fractions, nosotros are left with the following multiplication trouble:
$$ \frac{ane}{1} \times \frac{1}{4}$$
In fraction multiplication, we multiply all values on the top half of the fraction together and multiply all values on the bottom half of the fraction together:
$$ \frac{1}{1} \times \frac{1}{four} = \frac{ane \times 1}{1 \times four} = \frac{1}{four}$$
The last answer should exist ¼.
Division works in a like fashion to multiplication. In cases of sectionalisation, we multiply past the reciprocal of the divisor. For example, if we're dividing the post-obit fractions:
$$ \frac{1}{iii} \div \frac{4}{7}$$
Continue by multiplying the first fraction by the reciprocal of the second fraction (74). Then, nosotros tin proceed past the same rules as with multiplication:
$$ \frac{i}{3} \div \frac{iv}{7} = \frac{1}{3} \times \frac{7}{4} = \frac{1 \times vii}{3 \times 4} = \frac{7}{12}$$
The terminal reply should exist 7/12.
c) Application: sparse lens equation
The thin lens equation is a common application of fraction math and physics on the MCAT. The sparse lens equation relates different lengths associated with geometric optics: focal length, object distance, and paradigm distance. Focal length (f) refers to the distance betwixt the focal point and the center of a lens or mirror. Object length (o) refers to the altitude between the object and the center of the lens or mirror. Image distance (i) is the distance between the epitome and the center of the lens or mirror.
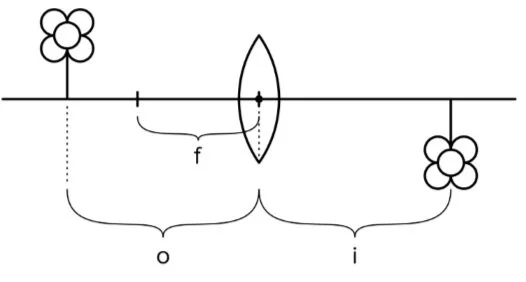
Figure: The sparse lens equation relates the distances f, i, and o.
The thin lens equation is as follows:
$$ \frac{i}{f} = \frac{1}{o} + \frac{1}{i}$$ $$ \mbox{where f = focal length,}$$ $$ \mbox{o = object distance,}$$ $$ \mbox{and i = image distance} $$
The sign of each variable contains information about its location.
-
Positive focal lengths are characteristic of concave mirrors and converging lenses; negative focal lengths are characteristic of convex mirrors and diverging lenses.
-
Positive object distances occur when the object is in front of the mirror or lens, and negative object distances occur when the object is behind. In nearly all applications relevant to the MCAT, the object distance is positive.
-
If the image altitude is positive, the image is in front of the mirror or backside a lens. Such an image is referred to every bit a existent image and is always inverted.
-
If the image distance is negative, the image is behind the mirror or in front of a lens. Such an epitome is a virtual image and is e'er upright.
Yous can observe more than information well-nigh mirrors and lenses in our guide on light and optics.
When dealing with algebraic equations like the thin lens equation, recollect to apply the same operations to both sides of the equation. For instance, if nosotros subtract 4 from one side of the equation, we must also subtract 4 from the other side of the equation. Doing and so keeps the equation counterbalanced. Failing to practise so may atomic number 82 yous to an incorrect respond!
d) Example trouble
Let's work on an application of the thin lens equation.
Suppose a converging lens with a focal length of 4 cm is placed half-dozen cm away from an object. Determine whether the prototype produced past this lens is real or virtual.
Get-go past writing downward an equation you already know: the thin lens equation itself.
$$ \frac{1}{f} = \frac{1}{o} + \frac{1}{i}$$
This question wants usa to determine the value of the image distance, or i. We tin isolate that variable on 1 side of the equation by subtracting ane/o from both sides of the equation.
$$ \frac{1}{i} = \frac{1}{f} - \frac{one}{o}$$
At present, we can evaluate the expression using our known values for f=4 cm and o=6 cm.
$$ \frac{1}{i} = \frac{1}{4}\space cm^{-1} - \frac{1}{6}\space cm^{-one}$$
To subtract these numbers, we rewrite these fractions with a common denominator. In this example, nosotros volition use 4x6 = 24.
$$ \frac{ane}{i} = \frac{6}{24}\infinite cm^{-1} - \frac{4}{24}\space cm^{-1}$$
Continuing to evaluate the expression:
$$ \frac{one}{i} = \frac{6-4}{24}\space cm^{-1}$$ $$ \frac{1}{i} = \frac{2}{24}\space cm^{-ane}$$
This brings us a bit closer to solving for i. To obtain an verbal value for i, nosotros will need to simplify these expressions without the use of fractions. Fortunately, we can cantankerous-multiply here: multiply the numerator of the fraction on the left-hand side by the denominator of the fraction on the right-hand side, and vice versa.
$$ 1 \times 24 = 2 \times i$$ $$ 24 = 2i $$ $$24 \div 2 = i $$ $$i = 12 \space cm $$
Because our image distance is positive (i > 0), we know that the image is real.
Acknowledgements: Sneha Mittal
Role 6: Passage-based questions and answers
In a college laboratory experiment, students were asked to complete a titration experiment with acerb acid as analyte and sodium hydroxide equally titrant. To deport the experiment, 100 mL of 0.200M NaOH was added to a burette. 50 mL of acetic acrid of unknown concentration was added to an Erlenmeyer flask and placed below the burette. Students were asked to determine the concentration of the provided acetic acrid (pKa = 4.seven).
1 of the students generates the post-obit titration curve while completing the assignment experiment.

Effigy 1: A student-generated titration bend.
Question 1: Approximately how many moles of NaOH were present in the burette at the equivalence point of the student'southward titration?
A) 0.03 mol
B) 0.029 mol
C) 0.014 mol
D) 0.01 mol
Question ii: How many grams of NaOH were added to 100 mL water to create the solution used in the titration?
A) 0.8 grand
B) 1.2 g
C) 0.5 yard
D) 0.three g
Question iii: What is the approximate ratio of [HA] to [A-] at the equivalence point of the titration?
A) 103.3
B) 104.three
C) 10-iv.3
D) 10-three.3
Question iv: Past how many orders of magnitude is the concentration of acetic acrid after the educatee has added fourteen mL NaOH greater than the concentration of acetic acid after the student has added 28 mL NaOH?
Question five: Assume that instead of acerb acrid, formic acid (pKa = iii.7) of the aforementioned concentration was used in this experiment. How would this affect the pH of the equivalence indicate?
A) The equivalence point would increase
B) The equivalence indicate would decrease
C) No change
D) Either A or C
Answers to passage-based questions
ane. Answer selection C is right. At equivalence point, 28 mL NaOH was added to the Erlenmeyer flask, leaving 72 mL in the burette (100mL – 28mL = 72 mL). To determine the number of moles in 72 mL NaOH, we comport the post-obit calculations.
$$ 72 \infinite mL \infinite \times \frac{1 \space 50}{1000 \space mL} \times 0.200 \space ^{mol}/_L =0.0144 \space mol$$
Given that 1 of the original numbers (72 mL) has ii significant figures, nosotros must too round our result to 2 significant figures. The right reply is 0.014 mol.
ii. Reply pick A is correct. Our calculations for this problem are similar to those from the final problem. In this case, we first with the full volume of NaOH mL and must also summate the number of grams in one mole of NaOH.
Na has an atomic mass of 22.99 grams/mol, which we will round to 23 grams/mol. O has an diminutive mass of 15.99 grams/mol, which we volition round to 16 grams/mol. H has an diminutive mass of ane.01 grams/mol, which we will round to 1 gram/mol.
Thus, the tooth mass of NaOH is 23 + 16 + one = 40 grand/mol.
At this phase, we tin use dimensional analysis and use the known molarity of the solution to calculate the number of grams NaOH added:
$$ 100 \space mL \times \frac{i \infinite Fifty}{1000 \space mL} \times 0.200 \space ^{mol}/_L \times 40 \space ^g/_{mol} = 0.eight \infinite m$$
3. Answer choice C is right. We can use the Henderson-Hasselbalch equation to solve for the ratio. At the equivalence indicate, the pH is approximately ix. The pKa of acerb acid is equal to 4.seven. This question asks for u.s. to notice the ratio of [HA] to [A-], or [HA]/[A-].
$$ pH = pK_a + log(\frac{[A^-]}{[HA]})$$ $$ pH-pK_a = log(\frac{[A^-]}{[HA]})$$ $$ -1 \times (pH - pK_a) = -ane \times log(\frac{[A^-]}{[HA]})$$ $$ pK_a - pH = log(\frac{[HA]}{[A^-]})$$ $$ 4.seven - 9 = log(\frac{[HA]}{[A^-]})$$ $$ -4.3 = log(\frac{[HA]}{[A^-]}$$ $$ 10^{-4.3} = \frac{[HA]}{[A^-]}$$
4. Answer choice D is correct. The pH effectually 14 mL NaOH is 2 and effectually 28 mL NaOH is 9. The difference between these numbers is vii, which represents the difference between the orders of magnitude of the two concentrations (selection D is correct). Answer C gives the exact ratio of these differences rather than the social club of magnitude.
v. Answer choice B is correct. To answer this question, we must call up that pKa is the –log(Ka); thus, the higher the Ga value, the smaller the pKa value. Since the pGa of formic acrid is less than that of acetic acid, its Thoua value is higher. Because formic acid is a stronger acid, it will have a weaker conjugate base, which volition not react as readily with water to produce formic acrid and hydroxide. Every bit a event of this decreased production of hydroxide, the pH of the solution will decrease (B).
Part 7: Standalone questions and answers
Question ane: Suppose that a solution in a chalice is noted to have the post-obit properties:
[HA] = 0.001, [A-] = 0.one and pKa = 3.2
What is the approximate pH of this solution?
A) ten-1.two
B) x-5.two
C) one.two
D) 5.2
Question 2: How many significant figures are in 2090.ten?
A) iv
B) 3
C) 5
D) 6
Question three: What is the number 909.90 written in scientific notation?
A) 9.099 x 104
B) 9.099 x 102
C) 9.0990 x xtwo
D) 9.0990 x 104
Question 4: Sound A is 200 decibels and Sound B is 40 decibels. By how many orders of magnitude is Sound A greater than Sound B?
A) 160
B) xvi
C) 5
D) 50
Question v: A student is asked to ready a 43.vi m sample of hypochlorous acid (HClO). How many moles of HClO should the student prepare?
A) 0.672 mol HOCl
B) 0.237 mol HOCl
C) 0.831 mol HOCl
D) one.20 mol HOCl
Answer fundamental for standalone questions:
-
Answer choice D is right. We can use the Henderson-Hasselbalch equation to solve for the pH. Three of the values used in the Henderson-Hasselbalch equation are given to u.s.a. directly in the question stem.
$$ pH = pK_a + log(\frac{[A^-]}{[HA]})$$ $$ pH = 3.ii + log(\frac{0.1}{0.001})$$ $$ pH = iii.2 + log(100) $$ $$ pH = 3.2 + 2 $$ $$ pH = five.2 $$
ii. Answer pick D is correct. Using the rules for significant figures, count all 3 digits to the left of the decimal signal as pregnant. We also know to count the 2 zeroes in the hundreds and ones places as pregnant because they are between two nonzero numbers. The cipher in the hundredths place is right of a nonzero number, making information technology significant too. This amounts to 6 significant figures.
three. Answer choice C is correct. In scientific notation, the absolute value of the significand is less than ten. Thus, we rewrite 909.90 past moving the decimal point to the left two places. To compensate for this adjustment, nosotros multiply the significand by a gene of 100. The only answer option that does this and retains the number of pregnant figures from the original notation is C.
4. Reply option B is correct. We tin answer this question using the equation to calculate decibels. To compare ii unlike sound levels, nosotros can calculate the intensity of each sound, then calculate the ratio of both intensities.
Here, nosotros volition denote the intensity of sound A equally IA and the intensity of sound B as IB. $$ dB = 10 log(\frac{I}{I_0}) $$ $$ 200 = 10 log(\frac{I_A}{I_0}) $$ $$ 20 = log(\frac{I_A}{I_0}) $$ $$ 10^{20} = \frac{I_A}{I_0} $$ $$ I_0 \times ten^{20} = I_A $$ $$\mbox{Then, } 40 = 10 log(\frac{I_B}{I_0})$$ $$ 4 = log(\frac{I_B}{I_0})$$ $$ ten^iv = \frac{I_B}{I_0} $$ $$ I_0 \times x^4 = I_B$$ $$ \mbox{Thus, } \frac{I_0 \times 10^{20}}{I_0 \times x^4} = \frac{I_A}{I_B} $$ $$ten^{xvi} = \frac{I_A}{I_B} $$
The ratio of IA/ IB is 10xvi, indicating that Sound A is xvi orders of magnitude greater than Sound B.
five. Respond choice C is correct. To determine the number of moles, we convert this given mass using the molar mass of HOCl. First, nosotros calculate the tooth mass of HOCl.
The molar mass of H is 1.01 thou/mol. We can round this to 1 g/mol.
The molar mass of O is 15.99 chiliad/mol. We can round this to 16 yard/mol.
The molar mass of HClO tin thus exist approximated as:
$$1 \space ^g/_{mol} \space H + 16 \space ^g/_{mol} \space O + 35.45 \infinite ^g/_{mol} \space Cl = 52.45 \space ^g/_{mol} \infinite HOCl $$
To calculate the number of moles in a sample with known mass, we must divide the mass by the molar mass.
$$43.6 \mbox{ g HOCl } \div (52.457 \space ^g/_{mol} \space HOCl)$$
At this signal, we can eliminate answer option D. 43.6 is less than the number it is being divided by (52.47), and so the answer to this division problem must be less than 1.
Since the answer choices are far apart from each other, nosotros can round down our numbers to the nearest ten. This will requite us a ratio that should be easier to summate.
$$40 \space g \space HOCl \div (50 \space ^g/_{mol} \space HOCl) = 0.8 \mbox{ mol HOCl}$$
This estimated answer is fairly close to 0.831 mol, given by answer pick C.
Source: https://www.shemmassianconsulting.com/blog/math-mcat
0 Response to "What Math Do You Need to Know for Astrophysics"
Enregistrer un commentaire